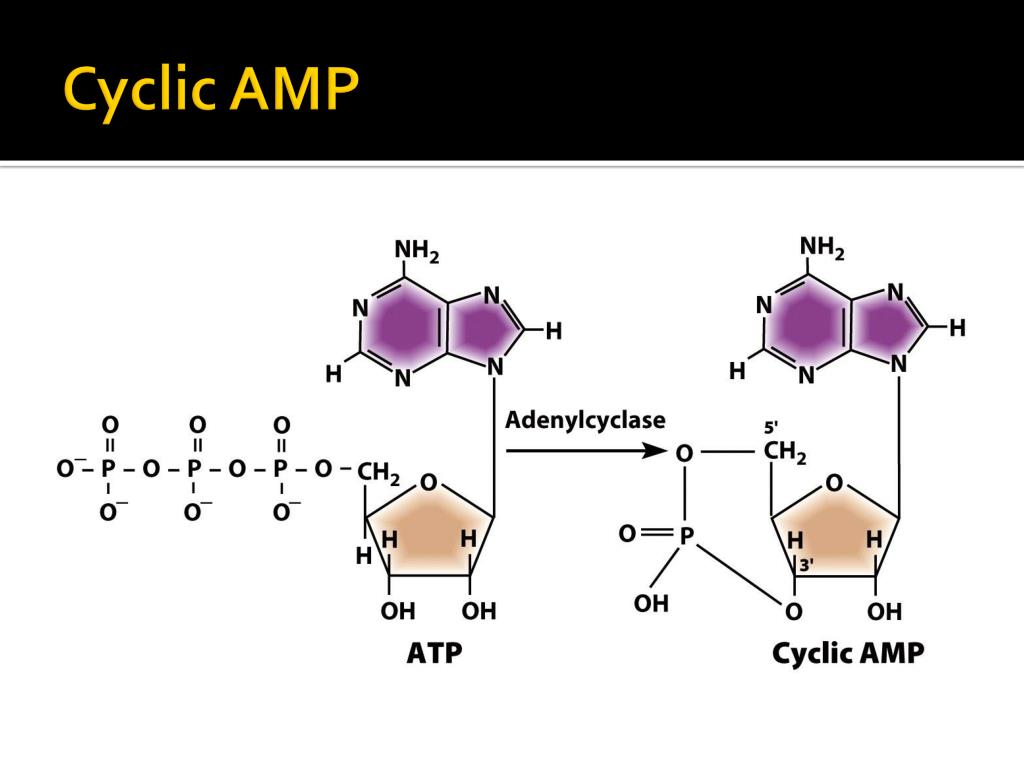
However, thermal instability of the biosensor may pose a problem for in vivo studies. As well, the GloSensor-22F biosensor had a superior signal-to-background ratio and the effect of temperature on biosensor activity was reversible. We found that preincubation and luminescence reading at room temperature were optimal as compared to higher temperatures. Temperature optimization studies were carried out using HEK293H cells transiently transfected with the adenosine receptor A2a and the GloSensor™ plasmid (pGloSensor-20F or -22F). 
We also present a detailed protocol for monitoring cAMP levels in live cells expressing endogenous or exogenous GPCRs. Here, we describe the temperature sensitivity and reversibility of the GloSensor™ cAMP assay, and which GloSensor™ version is optimal for measuring cytosolic cAMP. As well, it has not been reported whether the effects of temperature on biosensor activity are reversible. However, it is crucial to optimize assay conditions such as temperature. The GloSensor™ cAMP assay enables real-time monitoring of signaling downstream of many GPCRs. In this regard, luminescence-based biosensors have revolutionized our ability to monitor GPCR signaling kinetics. Monitoring intracellular cAMP levels can help identify orthosteric agonists and antagonists, as well as allosteric modulators. The second messenger cyclic adenosine monophosphate (cAMP) is a key mediator in many GPCR signaling pathways. G protein-coupled receptors (GPCRs) are an important receptor superfamily and common therapeutic targets.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
